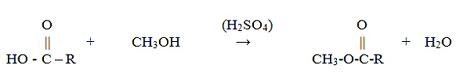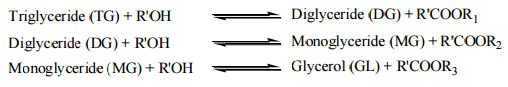group mini project
project 2
CLICK HERE TO DOWNLOAD THE FULL PROJECT
| krd_project_2.docx | |
| File Size: | 605 kb |
| File Type: | docx |
TESCO Seri Iskandar is selling a large volume of wet food, which mainly consisted of meat and fish section AND fruit and vegetables section. At the moment, we are not sure how they disposed the expired items. Nonetheless, as a chemical engineer, surely we can find many environmentally friendly, yet cost effective ways to ensure we can convert these waste into wealth.
In your team, you are required to figure out how you could solve these issues. Listed below are the requirement of your project:
1. Identify how much waste is being produced on daily basis (in kg or volume)
In your team, you are required to figure out how you could solve these issues. Listed below are the requirement of your project:
1. Identify how much waste is being produced on daily basis (in kg or volume)
Vegetable oil always believed to be as a substitute for fossil fuel product, diesel ever since in the early 19th century by the inventor of biodiesel engines, Rudolf Christian Karl Diesel. Seed oil is unconventional because to produce biodiesel, oils are extracted from vegetables which could have been used for conventional uses. In order to design a feasible method of biodiesel production, we used waste cooking oil which is used palm oil from cooking. The waste was being produced from a local restaurant from Seri Iskandar, Perak location and since Malaysia has a very large industrialization on palm oil production. This fact is advantage since the properties of WCO obtained from almost many regions in Malaysia would have same properties as this provides green light for Malaysia to be largest Biodiesel producer followed by United States and North America [1].
Waste cooking oil which is sourced from the local restaurant is 5 kilogram of used oil. The restaurants purchases cooking palm oil for frying and cooking purposes for her restaurant. To get precise information, the said restaurant purchases a famous “KNIFE BRAND” cooking palm oil, where most of the restaurants uses such brands. The waste cooking oil was mostly containing oleic acid and n-hexadeconic acid. There are a large difference in properties of unused cooking oil and used cooking oil values in cooking palm oil and when it comes to biodiesel production, values like density could affect the calculations designing for reactor (mass balance). Based on local researchers and the density of used cooking oil values from the same location, Seri Iskandar , Perak is 0.9013 g/cm3 [2].
Waste cooking oil which is sourced from the local restaurant is 5 kilogram of used oil. The restaurants purchases cooking palm oil for frying and cooking purposes for her restaurant. To get precise information, the said restaurant purchases a famous “KNIFE BRAND” cooking palm oil, where most of the restaurants uses such brands. The waste cooking oil was mostly containing oleic acid and n-hexadeconic acid. There are a large difference in properties of unused cooking oil and used cooking oil values in cooking palm oil and when it comes to biodiesel production, values like density could affect the calculations designing for reactor (mass balance). Based on local researchers and the density of used cooking oil values from the same location, Seri Iskandar , Perak is 0.9013 g/cm3 [2].
2. Conduct a literature survey of how these waste can be turned into wealth opportunity. Specify:
a. The varieties of available technologies
b. The reactions involved
c. The researchers or practitioners that made these technologies successful in their local area
d. The pros and constraints of these technologies
e. The way forward that would help your project
a. The varieties of available technologies
b. The reactions involved
c. The researchers or practitioners that made these technologies successful in their local area
d. The pros and constraints of these technologies
e. The way forward that would help your project
a. The varieties of available technologies
2.1-1 Mechanical stirring method
In modern industrial biodiesel process, the conversion of Vegetable oil or waste cooking oil to FAME achieved using homogenous catalyst system in batch or continuous mode through mechanical stirring method at the temperature 60 -70 degree Celsius and process comes to completion after 90 minutes.[3]
2.1-2 Microwave method
A far better concept of mixing in production of biodiesel in batch or continuous via heating of raw material through convection, conduction and radiation. Microwave irradiation is conventional method to enhance hydrolysis, esterification, and transesterification due to economical and convenient aspects.[4]
2.1-3 Ultrasonic Method
Ultrasonic is a very desirable tool for producing biodiesel from vegetable oil and animal fats, because it lowers the cost of processing, speeds up transesterification, does not require elevated temperatures, and produces a higher grade of biodiesel. . Although this phenomenon, known as cavitation, lasts but a few microseconds, and the amount of energy released by each bubble is minimal, the cumulative amount of energy generated is extremely high. [5]
b. The reaction involved
2.2-1 Esterification Process
Esterification, as it applies to biodiesel production, is the chemical reaction by which a fatty acid, typically a free fatty acid in degraded or second-use oil, reacts with an alcohol to produce an alkyl ester and water. The process differs from the transesterification reaction in that the reaction is occurring directly between the alcohol and the fatty acid molecule. The intermediate steps of cleaving the fatty acid chains from the glycerine backbone are not present. For this reason, no glycerine is produced during the esterification reaction. [6]
2.1-1 Mechanical stirring method
In modern industrial biodiesel process, the conversion of Vegetable oil or waste cooking oil to FAME achieved using homogenous catalyst system in batch or continuous mode through mechanical stirring method at the temperature 60 -70 degree Celsius and process comes to completion after 90 minutes.[3]
2.1-2 Microwave method
A far better concept of mixing in production of biodiesel in batch or continuous via heating of raw material through convection, conduction and radiation. Microwave irradiation is conventional method to enhance hydrolysis, esterification, and transesterification due to economical and convenient aspects.[4]
2.1-3 Ultrasonic Method
Ultrasonic is a very desirable tool for producing biodiesel from vegetable oil and animal fats, because it lowers the cost of processing, speeds up transesterification, does not require elevated temperatures, and produces a higher grade of biodiesel. . Although this phenomenon, known as cavitation, lasts but a few microseconds, and the amount of energy released by each bubble is minimal, the cumulative amount of energy generated is extremely high. [5]
b. The reaction involved
2.2-1 Esterification Process
Esterification, as it applies to biodiesel production, is the chemical reaction by which a fatty acid, typically a free fatty acid in degraded or second-use oil, reacts with an alcohol to produce an alkyl ester and water. The process differs from the transesterification reaction in that the reaction is occurring directly between the alcohol and the fatty acid molecule. The intermediate steps of cleaving the fatty acid chains from the glycerine backbone are not present. For this reason, no glycerine is produced during the esterification reaction. [6]
Figure 1: Esterification Process
2.2-2 Transesterification Process
The final step of obtaining Fatty Acid Methyl Ester (FAME) by reacting with alcohol such as methanol and Ethanol with triglyceride molecules is knows as transesterification. This process is relatively slow under optimum conditions during in absence of catalyst. For transesterification process , an alkaline catalyst such as KOH ( Potassium Hydroxide) or NaOH ( sodium hydroxide) to elevate the reaction at STP and to produce FAME and glycerol after step by step conversion as shown below. [7]
The final step of obtaining Fatty Acid Methyl Ester (FAME) by reacting with alcohol such as methanol and Ethanol with triglyceride molecules is knows as transesterification. This process is relatively slow under optimum conditions during in absence of catalyst. For transesterification process , an alkaline catalyst such as KOH ( Potassium Hydroxide) or NaOH ( sodium hydroxide) to elevate the reaction at STP and to produce FAME and glycerol after step by step conversion as shown below. [7]
Figure 2: Transesterification Process
c. The researchers or practitioners that made these technologies successful in their local area
There are few researches from both internationally and nationally who have been successful in showing their technologies to the world with both industrial and future technology aspects.
The method mechanical stirring was studied from two researchers. The method feasibility was studied from Bio resource Technology journal under a review of Biodiesel production from Fangrui Ma and Milford A Hanna [3], where various method was studied such as Blending (mechanical stirring), thermal cracking and transesterification.
Where else, the other technology which is ultrasonification was studied from few researchers as it was showing a promise reaction over short time and high yield. The researchers who have provided viable information for this method was Egi Augustin[8] on the research paper; production of biodiesel from waste cooking oil using ultrasonic tubular reactor. The next researcher who have achieved in this technology is Georgene Elizabeth Grant and Veera Gnaneswar Gude on the research study of; Kinetics of ultrasonic transesterification of waste cooking oil [9]. At last, the research followed to complete this project is; utilizing ultrasonic energy for reduction of free fatty acids in crude palm oil by [10].
It has to be remarked that two out of three research papers referred for ultrasonic technology was obtained from local Malaysian researchers.
d. The pros and constraints of these technologies
The mechanical stirring method is widely used in today's process with advantages of convenient, easy to repair and efficient physical mixing over the time. This mechanical stirring method in production of FAME has efficiency defect in time and power consumption aspect where it consumes 120 minutes for conversion and over the reaction, accumulation of soap and other constituents might interfere during stirring and inhibit the FAME production.
The microwave method is advantageous in the aspect of its convenience, rapidity, and economy advances in equipment design, and trends in electrical energy costs. Whereas the constraints of this technology is the amount of reactants feed could be higher due to radiation where in both esterification and transesterification excess methanol is needed.
Ultrasonification method is famous latest method where many researches acquiring the use of this technology for high intensity mixing due to cavitation and particularly in FAME production, it helps to prevent the formation of soap. The disadvantage of this technology is unavailability of large size probe
e. The way forward that would help your project
These technologies according to the advantages and disadvantages, the magnetic stirring which is the usually used in industries has relatively consumes larger power and more time to achieve biodiesel conversion. Where else, the ultrasonification method is new era process where currently the biodiesel conversion via ultrasonic, yet to be commercialized. The key factor for the reaction involving is the mixing of reactant and catalyst at optimum temperature. Hence, observing on all of the technologies, ultrasonification on both esterification and transesterification shows promising reaction at very short time. The technology of cavitation increases the mixing intensity and yield of biodiesel (FAME).
3. Identify the technology that you’d like to propose:-
a. What is it?
b. Why do you choose this technology? Why not others?
c. Is it sustainable technology?
d. Is there any catalyst involved? What type of catalyst? How much is required?
a. What is it?
b. Why do you choose this technology? Why not others?
c. Is it sustainable technology?
d. Is there any catalyst involved? What type of catalyst? How much is required?
a. What is it?
Ultrasonic is a very desirable tool for producing biodiesel from vegetable oil and animal fats, because it lowers the cost of processing, speeds up transesterification, does not require elevated temperatures, and produces a higher grade of biodiesel. The longitudinal vibrations of the ultrasonic probe are transmitted into the liquid as ultrasonic waves consisting of alternate expansions and compressions. The pressure fluctuations give birth to microscopic bubbles (cavities) which expand during the negative pressure excursions, and implode violently during the positive excursions. As the bubbles collapse, millions of shock waves eddy, and extremes in pressure and temperature are generated at the implosion sites. Although this phenomenon, known as cavitation, lasts but a few microseconds, and the amount of energy released by each bubble is minimal, the cumulative amount of energy generated is extremely high. [5]
b. Why do you choose this technology? Why not others?
According to all the technology proposed, Ultrasonic technology for producing higher yield of biodiesel due to alternate expansions and compression compared to other method such as mechanical stirring and microwave irradiation. Mixing of reactants at high speed is needed and only could be possible with ultrasonic. With high pressure and violent implode due to cavitation at high intensity favoured over other technologies. For example, the stirring method takes minimum 7 hours to obtain first level FAME conversion while microwave irradiation uses high power through conduction, convection and radiation for increased mixing intensity.
c. Is it sustainable technology?
According to the comparison study of mechanical stirring and ultrasonic [11] for biodiesel production, the power used for conventional process in industry yields 89% where ultrasonic yields 95% with less power consumption. The conventional method also consumes 120 minutes where ultrasonic technology takes up to only 5 minutes for FAME conversion. According to these key points, since less power consumption the economic shows promising growth and with less time reaction both the environment and social prevails its sustainability.
d. Is there any catalyst involved? What type of catalyst? How much is required?
There is catalyst involved during reaction of conversion from waste cooking oil to biodiesel or also known as FAME (Fatty Acid Methyl Ester). For the reaction in first place which is esterification uses Sulphuric Acid as catalyst and the amount used is 7 % wt [12]. This is the first process to obtain esterified product to undergo transesterification process for complete Biodiesel product.
Where else, in the second reaction which transesterification, the catalyst used for biodiesel conversion is Potassium Hydroxide which yields FAME and glycerol along with trace amount of water and soap. The proposed amount of catalyst is 1.5% wt [13]
Ultrasonic is a very desirable tool for producing biodiesel from vegetable oil and animal fats, because it lowers the cost of processing, speeds up transesterification, does not require elevated temperatures, and produces a higher grade of biodiesel. The longitudinal vibrations of the ultrasonic probe are transmitted into the liquid as ultrasonic waves consisting of alternate expansions and compressions. The pressure fluctuations give birth to microscopic bubbles (cavities) which expand during the negative pressure excursions, and implode violently during the positive excursions. As the bubbles collapse, millions of shock waves eddy, and extremes in pressure and temperature are generated at the implosion sites. Although this phenomenon, known as cavitation, lasts but a few microseconds, and the amount of energy released by each bubble is minimal, the cumulative amount of energy generated is extremely high. [5]
b. Why do you choose this technology? Why not others?
According to all the technology proposed, Ultrasonic technology for producing higher yield of biodiesel due to alternate expansions and compression compared to other method such as mechanical stirring and microwave irradiation. Mixing of reactants at high speed is needed and only could be possible with ultrasonic. With high pressure and violent implode due to cavitation at high intensity favoured over other technologies. For example, the stirring method takes minimum 7 hours to obtain first level FAME conversion while microwave irradiation uses high power through conduction, convection and radiation for increased mixing intensity.
c. Is it sustainable technology?
According to the comparison study of mechanical stirring and ultrasonic [11] for biodiesel production, the power used for conventional process in industry yields 89% where ultrasonic yields 95% with less power consumption. The conventional method also consumes 120 minutes where ultrasonic technology takes up to only 5 minutes for FAME conversion. According to these key points, since less power consumption the economic shows promising growth and with less time reaction both the environment and social prevails its sustainability.
d. Is there any catalyst involved? What type of catalyst? How much is required?
There is catalyst involved during reaction of conversion from waste cooking oil to biodiesel or also known as FAME (Fatty Acid Methyl Ester). For the reaction in first place which is esterification uses Sulphuric Acid as catalyst and the amount used is 7 % wt [12]. This is the first process to obtain esterified product to undergo transesterification process for complete Biodiesel product.
Where else, in the second reaction which transesterification, the catalyst used for biodiesel conversion is Potassium Hydroxide which yields FAME and glycerol along with trace amount of water and soap. The proposed amount of catalyst is 1.5% wt [13]
4. Mass Balance
There are two reaction involved in the production of waste cooking oil to biodiesel which are esterification and transesterification via ultrasonification.
For esterification the basic properties used are waste cooking oil of 5kg and the alcohol to oil ratio used was 20:1 [12]. The general equation for esterification is ;
[Waste Cooking Oil] + [methanol] = [methyl ester] + [ water] + [ methanol] + [ oil]
5.9[Waste Cooking Oil] + 118 [methanol] =5.78 [methyl ester] + 5.78[ water] + 112.22[ methanol] + 0.12[ oil]
Therefore, the feed of 5kg with the reaction with methanol and catalyst (H2SO4) amount 7wt% yields with 98% [cite] to esterified methyl ester to transesterification process with output of 4.65kg.(referring tripalmitin)[14]
For transesterification process of reacting methyl ester with alcohol with alcohol:oil molar ratio of 6:1 in the presence of catalyst (NaOH) of 1.5wt %[cite] with 98% efficiency according to below general equation;
[Triglyceride] + 3 [ methanol] = [Fat acid methyl ester] + [ glycerol]
5.78[Triglyceride] + 34.68[ methanol] = 5.66[Fat acid methyl ester] + 5.66 [ glycerol] + 0.12[ triglyceride] + 17.34 [ methanol]
Therefore for the feed of 4.65 kg of Tripalmitin [14] under transesterification produces 3.8kg of biodiesel which is fatty acid methyl palmitate. [14]
References and citations
For esterification the basic properties used are waste cooking oil of 5kg and the alcohol to oil ratio used was 20:1 [12]. The general equation for esterification is ;
[Waste Cooking Oil] + [methanol] = [methyl ester] + [ water] + [ methanol] + [ oil]
5.9[Waste Cooking Oil] + 118 [methanol] =5.78 [methyl ester] + 5.78[ water] + 112.22[ methanol] + 0.12[ oil]
Therefore, the feed of 5kg with the reaction with methanol and catalyst (H2SO4) amount 7wt% yields with 98% [cite] to esterified methyl ester to transesterification process with output of 4.65kg.(referring tripalmitin)[14]
For transesterification process of reacting methyl ester with alcohol with alcohol:oil molar ratio of 6:1 in the presence of catalyst (NaOH) of 1.5wt %[cite] with 98% efficiency according to below general equation;
[Triglyceride] + 3 [ methanol] = [Fat acid methyl ester] + [ glycerol]
5.78[Triglyceride] + 34.68[ methanol] = 5.66[Fat acid methyl ester] + 5.66 [ glycerol] + 0.12[ triglyceride] + 17.34 [ methanol]
Therefore for the feed of 4.65 kg of Tripalmitin [14] under transesterification produces 3.8kg of biodiesel which is fatty acid methyl palmitate. [14]
References and citations
| reference.docx | |
| File Size: | 13 kb |
| File Type: | docx |
5. Design the reactor that will be used in the proposed technology.
a. What is the type of reactor to be used?
b. What is the size of the reactor?
c. Operating temperature and pressure?
d. Does it require any heating/cooling? How do you provide that?
e. Produce a mechanical drawing of the reactor
f. Cost of the reactor
a. What is the type of reactor to be used?
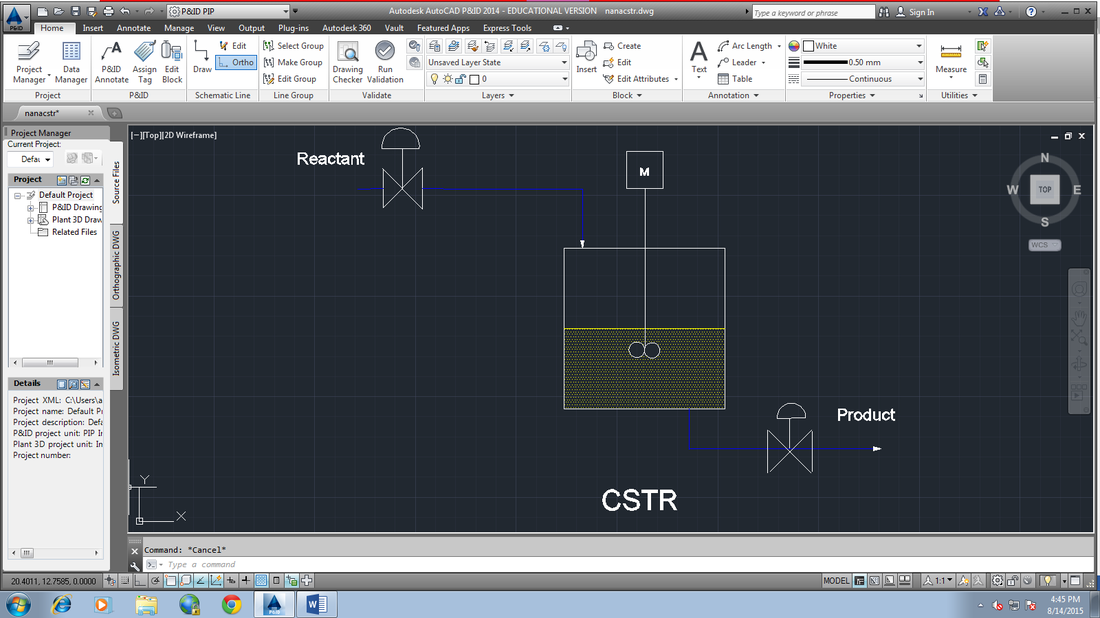
We will be using Continuous-stirred Tank Reactor (CSTR). In CSTR, any materials is easy and free to enter and exit. This reactor also operates at steady state.
The conditions do not change with time which will make our process conditions constant and not always changing which is easier to handle and control. The products are removed continuously and the reactants are entered into the reactor continuously.
The conditions of entering and exit streams are the same. CSTR are well mixed, the contents have uniform and equal properties such as the temperature, density and pressure. The volume of CSTR are usually at constant volume. There are stirrers called agitators inside CSTR to mix well the reactants and catalyst. A CSTR can act as loop reactor when heated. The pressurized fluid is inserted into the system to facilitate the stirring or the agitators for higher heat and mass transfer rates.
b. What is the size of the reactor?
a. What is the type of reactor to be used?
b. What is the size of the reactor?
c. Operating temperature and pressure?
d. Does it require any heating/cooling? How do you provide that?
e. Produce a mechanical drawing of the reactor
f. Cost of the reactor
a. What is the type of reactor to be used?
We will be using Continuous-stirred Tank Reactor (CSTR). In CSTR, any materials is easy and free to enter and exit. This reactor also operates at steady state.
The conditions do not change with time which will make our process conditions constant and not always changing which is easier to handle and control. The products are removed continuously and the reactants are entered into the reactor continuously.
The conditions of entering and exit streams are the same. CSTR are well mixed, the contents have uniform and equal properties such as the temperature, density and pressure. The volume of CSTR are usually at constant volume. There are stirrers called agitators inside CSTR to mix well the reactants and catalyst. A CSTR can act as loop reactor when heated. The pressurized fluid is inserted into the system to facilitate the stirring or the agitators for higher heat and mass transfer rates.
b. What is the size of the reactor?
| sizing-cstr.docx | |
| File Size: | 16 kb |
| File Type: | docx |
c. Operating temperature and pressure?
Temperature= 60 degree celcius
Pressure= 1 atm
d. Does it require any heating/cooling?How do you provide that?
The esterification and transesterification needs heating because heating can increases the rate of the reaction. Esterification of an ester is an equilibrium reaction, so one often uses an acid catalyst. It drives the reaction to completion.
e. Produce a mechanical drawing of the reactor
Temperature= 60 degree celcius
Pressure= 1 atm
d. Does it require any heating/cooling?How do you provide that?
The esterification and transesterification needs heating because heating can increases the rate of the reaction. Esterification of an ester is an equilibrium reaction, so one often uses an acid catalyst. It drives the reaction to completion.
e. Produce a mechanical drawing of the reactor
f. Cost of the reactor
| cost-cstr.docx | |
| File Size: | 13 kb |
| File Type: | docx |
6. Include the control element that you need to install in operating the reactor and specify your justification
a. What is the transfer function for the reactor?
b. What is the order of the transfer function?
c. Identify the control variable(s), manipulated variable(s), and possible disturbance variable(s) for the reactor.
d. What type of control algorithm (P, PI, or PID controllers) would you use? Why?
a. What is the transfer function for the reactor?
b. What is the order of the transfer function?
c. Identify the control variable(s), manipulated variable(s), and possible disturbance variable(s) for the reactor.
d. What type of control algorithm (P, PI, or PID controllers) would you use? Why?
| q6._control_system.docx | |
| File Size: | 26 kb |
| File Type: | docx |
7. Conduct Hazard and Operability Studies (HAZOP) of the reactor to identify any special safety features to be installed in your proposed reactor.
Hazard and Operability (HAZOP)
HAZOP is a study that identifies hazard and operability problems. HAZOP is done to investigate how the plant might deviate from design intent. During a HAZOP study, the solution becomes apparent for the problems that may incur, it is recorded as part of HAZOP result. Nevertheless we must ensure not to avoid trying to find solutions which are not so apparent, as the main objective for the HAZOP is problem identification. HAZOP is basically for safety, hazards are the main concern and operability problems degrade plant performance (product quality, production rate, profit). Hence, considerable engineering insight is required as engineers working independently could develop different results.
In our reactor, the main trancesterification reaction section was chosen as the subject of the HAZOP’s study because of its importance to the biodiesel plant. A hazard matrix was constructed to perform the hazard each unit operation. For unit operation, the process parameters such as flows, pressures, and temperatures were examined to determine possible deviations which could lead to definitive hazards. For each deviance, the HAZOPs study recognized all the probable causes for that deviation. The consequences for each deviation were then listed, and recommendations were made to minimalize the effect or to avert the consequences.
HAZARD MATRIX
PROCESS: TRANSESTERIFICATION REACTION
*attached below Matrix hazard
Hazard and Operability (HAZOP)
HAZOP is a study that identifies hazard and operability problems. HAZOP is done to investigate how the plant might deviate from design intent. During a HAZOP study, the solution becomes apparent for the problems that may incur, it is recorded as part of HAZOP result. Nevertheless we must ensure not to avoid trying to find solutions which are not so apparent, as the main objective for the HAZOP is problem identification. HAZOP is basically for safety, hazards are the main concern and operability problems degrade plant performance (product quality, production rate, profit). Hence, considerable engineering insight is required as engineers working independently could develop different results.
In our reactor, the main trancesterification reaction section was chosen as the subject of the HAZOP’s study because of its importance to the biodiesel plant. A hazard matrix was constructed to perform the hazard each unit operation. For unit operation, the process parameters such as flows, pressures, and temperatures were examined to determine possible deviations which could lead to definitive hazards. For each deviance, the HAZOPs study recognized all the probable causes for that deviation. The consequences for each deviation were then listed, and recommendations were made to minimalize the effect or to avert the consequences.
HAZARD MATRIX
PROCESS: TRANSESTERIFICATION REACTION
*attached below Matrix hazard
| hazop.docx | |
| File Size: | 19 kb |
| File Type: | docx |
8. Draw P&ID diagram of the complete process.
| question_8.docx | |
| File Size: | 384 kb |
| File Type: | docx |